Comprehensive tutorial on ECG interpretation, covering normal waves, durations, intervals, rhythm and abnormal findings. From basic to advanced ECG reading. Includes a complete e-book, video lectures, clinical management, guidelines and much more. Importance of Lead aVL in STEMI Recognition. In 2013 an article was published in Prehospital Emergency Care that measured the ability of paramedics to identify acute STEMI on the 12-lead ECG. The results showed that paramedic accuracy varied depending on the infarct location. Inferior: 96%.
| Electrocardiography in myocardial infarction | |
|---|---|
| Medical diagnostics | |
12-lead electrocardiogram showing ST-segment elevation (orange) in I, aVL and V1-V5 with reciprocal changes (blue) in the inferior leads, indicative of an anterior wall myocardial infarction. | |
| Purpose | detecting ischemia or acute coronary injury in emergency department |
Electrocardiography in suspected myocardial infarction has the main purpose of detecting ischemia or acute coronary injury in emergency department populations coming for symptoms of myocardial infarction (MI). Also, it can distinguish clinically different types of myocardial infarction.
- 2Main patterns
Technical issues[edit]
The standard 12 lead electrocardiogram (ECG) has several limitations. An ECG represents a brief sample in time. Because unstable ischemic syndromes have rapidly changing supply versus demand characteristics, a single ECG may not accurately represent the entire picture.[1] It is therefore desirable to obtain serial 12 lead ECGs, particularly if the first ECG is obtained during a pain-free episode. Alternatively, many emergency departments and chest pain centers use computers capable of continuous ST segment monitoring.[2] The standard 12 lead ECG also does not directly examine the right ventricle, and is relatively poor at examining the posterior basal and lateral walls of the left ventricle. In particular, acute myocardial infarction in the distribution of the circumflex artery is likely to produce a nondiagnostic ECG.[1] The use of additional ECG leads like right-sided leads V3R and V4R and posterior leads V7, V8, and V9 may improve sensitivity for right ventricular and posterior myocardial infarction. In spite of these limitations, the 12 lead ECG stands at the center of risk stratification for the patient with suspected acute myocardial infarction. Mistakes in interpretation are relatively common, and the failure to identify high risk features has a negative effect on the quality of patient care.[3]
Main patterns[edit]
The 12 lead ECG is used to classify MI patients into one of three groups:[4]
- those with ST segment elevation or new bundle branch block (suspicious for acute injury and a possible candidate for acute reperfusion therapy with thrombolytics or primary PCI),
- those with ST segment depression or T wave inversion (suspicious for ischemia), and
- those with a so-called non-diagnostic or normal ECG. However, a normal ECG does not rule out acute myocardial infarction.
ST elevation MI[edit]
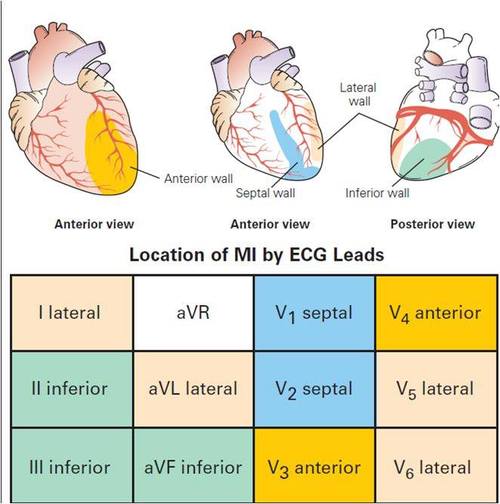
The 2018 European Society of Cardiology/American College of Cardiology Foundation/American Heart Association/World Health Federation Universal Definition of Myocardial Infarction for the ECG diagnosis of the ST segment elevation type of acute myocardial infarction require new ST elevation at J point of at least 1mm (0.1 mV) in two contiguous leads with the cut-points: ≥1 mm in all leads other than leads V2-V3. For leads V2-V3: ≥2 mm in men ≥40 years, ≥2.5 mm in men <40 years, or ≥1.5 mm in women regardless of age. This assumes usual calibration of 1mV/10mm.[5] These elevations must be present in anatomically contiguous leads.[4] (I, aVL, V5, V6 correspond to the lateral wall; V3-V4 correspond to the anterior wall ; V1-V2 correspond to the septal wall; II, III, aVF correspond to the inferior wall.) This criterion is problematic, however, as acute myocardial infarction is not the most common cause of ST segment elevation in chest pain patients.[6] Over 90% of healthy men have at least 1 mm (0.1 mV) of ST segment elevation in at least one precordial lead.[7] The clinician must therefore be well versed in recognizing the so-called ECG mimics of acute myocardial infarction, which include left ventricular hypertrophy, left bundle branch block, paced rhythm, early repolarization, pericarditis, hyperkalemia, and ventricular aneurysm.[7][8][9]
There are heavily researched clinical decision tools such as the TIMI Scores which help prognose and diagnose STEMI based on clinical data. For example, TIMI scores are frequently used to take advantage of EKG findings for prognosing patients with MI symptoms.[10] Based on symptoms and electrocardiographic findings, practitioners can differentiate between unstable angina, NSTEMI and STEMI, normally in the emergency room setting.[11] Other calculators such as the GRACE[12] and HEART [13] scores, assess other major cardiac events using electrocardiogram findings, both predicting mortality rates for 6 months and 6 weeks, respectively.[citation needed]
Typical progression[edit]
Sometimes the earliest presentation of acute myocardial infarction is the hyperacute T wave, which is treated the same as ST segment elevation.[14] In practice this is rarely seen, because it only exists for 2–30 minutes after the onset of infarction.[15] Hyperacute T waves need to be distinguished from the peaked T waves associated with hyperkalemia.[16]
In the first few hours the ST segments usually begin to rise.[17] Pathological Q waves may appear within hours or may take greater than 24 hr.[17] The T wave will generally become inverted in the first 24 hours, as the ST elevation begins to resolve.[17]
Long term changes of ECG include persistent Q waves (in 90% of cases) and persistent inverted T waves.[17] Persistent ST elevation is rare except in the presence of a ventricular aneurysm.[17]
See also[edit]
References[edit]
- ^ abCannon CP at al. Management of Acute Coronary Syndromes. p. 175. New Jersey: Humana Press, 1999. ISBN0-89603-552-2.
- ^Selker HP, Zalenski RJ, Antman EM, et al. (January 1997). 'An evaluation of technologies for identifying acute cardiac ischemia in the emergency department: executive summary of a National Heart Attack Alert Program Working Group Report'. Ann Emerg Med. 29 (1): 1–12. doi:10.1016/S0196-0644(97)70297-X. PMID8998085.
- ^Masoudi FA, Magid DJ, Vinson DR, et al. (October 2006). 'Implications of the failure to identify high-risk electrocardiogram findings for the quality of care of patients with acute myocardial infarction: results of the Emergency Department Quality in Myocardial Infarction (EDQMI) study'. Circulation. 114 (15): 1565–71. doi:10.1161/CIRCULATIONAHA.106.623652. PMID17015790.
- ^ ab'2005 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care — Part 8: Stabilization of the Patient With .......Acute Coronary Syndromes'. Circulation. 112 (24_suppl): IV–89–IV–110. 2005. doi:10.1161/CIRCULATIONAHA.105.166561. PMID16314375.
- ^Thygesen, Kristian; Alpert, Joseph S.; Jaffe, Allan S.; Chaitman, Bernard R.; Bax, Jeroen J.; Morrow, David A.; White, Harvey D. (October 2018). 'Fourth Universal Definition of Myocardial Infarction (2018)'. Journal of the American College of Cardiology. 72 (18): 2252. doi:10.1016/j.jacc.2018.08.1038.
- ^Brady WJ, Perron AD, Martin ML, Beagle C, Aufderheide TP (January 2001). 'Cause of ST segment abnormality in ED chest pain patients'. Am J Emerg Med. 19 (1): 25–8. doi:10.1053/ajem.2001.18029. PMID11146012.
- ^ abWang K, Asinger RW, Marriott HJ (November 2003). 'ST-segment elevation in conditions other than acute myocardial infarction'. N. Engl. J. Med. 349 (22): 2128–35. doi:10.1056/NEJMra022580. PMID14645641.
- ^Brady WJ, Chan TC, Pollack M (January 2000). 'Electrocardiographic manifestations: patterns that confound the EKG diagnosis of acute myocardial infarction-left bundle branch block, ventricular paced rhythm, and left ventricular hypertrophy'. J Emerg Med. 18 (1): 71–8. doi:10.1016/S0736-4679(99)00178-X. PMID10645842.
- ^Brady WJ, Perron AD, Chan T (April 2001). 'Electrocardiographic ST-segment elevation: correct identification of acute myocardial infarction (AMI) and non-AMI syndromes by emergency physicians'. Acad Emerg Med. 8 (4): 349–60. doi:10.1111/j.1553-2712.2001.tb02113.x. PMID11282670.
- ^David A. Morrow; Elliott M. Antman; Andrew Charlesworth; Richard Cairns; Sabina A. Murphy; James A. de Lemos; Robert P. Giugliano; Carolyn H. McCabe; Eugene Braunwald (2000). 'TIMI Risk Score for ST-Elevation Myocardial Infarction: A Convenient, Bedside, Clinical Score for Risk Assessment at Presentation: An Intravenous nPA for Treatment of Infarcting Myocardium Early II Trial Substudy'. Circulation. 102 (17): 2031–7. doi:10.1161/01.cir.102.17.2031. PMID11044416.
- ^David A. Morrow; Elliott M. Antman; Andrew Charlesworth; Richard Cairns; Sabina A. Murphy; James A. de Lemos; Robert P. Giugliano; Carolyn H. McCabe; Eugene Braunwald (2000). 'The TIMI Risk Score for Unstable Angina/Non–ST Elevation MI: A Method for Prognostication and Therapeutic Decision Making'. JAMA. 284 (7): 835–42. doi:10.1001/jama.284.7.835. PMID10938172.
- ^Fox KA, Dabbous OH, Goldberg RJ, Pieper KS, Eagle KA, Van de Werf F, Avezum A, Goodman SG, Flather MD, Anderson FA Jr, Granger CB (2006). 'Prediction of risk of death and myocardial infarction in the six months after presentation with acute coronary syndrome: prospective multinational observational study (GRACE)'. BMJ. 333 (7578): 2153–8. doi:10.1136/bmj.38985.646481.55. PMC1661748. PMID17032691.
- ^Backus BE, Six AJ, Kelder JC, Bosschaert MA, Mast EG, Mosterd A, Veldkamp RF, Wardeh AJ, Tio R, Braam R, Monnink SH, van Tooren R, Mast TP, van den Akker F, Cramer MJ, Poldervaart JM, Hoes AW, Doevendans PA (2013). 'A prospective validation of the HEART score for chest pain patients at the emergency department'. Circulation. 168 (3): 2153–8. doi:10.1016/j.ijcard.2013.01.255. PMID23465250.
- ^Somers MP, Brady WJ, Perron AD, Mattu A (May 2002). 'The Prominent T wave: electrocardiographic differential diagnosis'. Am J Emerg Med. 20 (3): 243–51. doi:10.1053/ajem.2002.32630. PMID11992348.
- ^Smith SW, Whitwam W (February 2006). 'Acute coronary syndromes'. Emerg. Med. Clin. North Am. 24 (1): 53–89, vi. doi:10.1016/j.emc.2005.08.008. PMID16308113.
- ^Van Mieghem C, Sabbe M, Knockaert D (April 2004). 'The clinical value of the ECG in noncardiac conditions'. Chest. 125 (4): 1561–76. doi:10.1378/chest.125.4.1561. PMID15078775. Archived from the original on 2008-12-10.
- ^ abcdegpnotebook.co.uk > ECG changes in myocardial infarction Retrieved on June 16, 2010
External links[edit]
- TIMI Risk Score for UA/NSTEMI and STEMI
Abstract
The clinical presentation of posterior myocardial infarction is not always easy, not even for the cardiologist. In this article a 70-year-old woman who presented with chest pain is described. The electrocardiogram at presentation showed marked ST-segment depression in leads V1 to V5 and slight ST-segment depression in leads I and aVL. There was ST-segment elevation in the posterior leads V7 to V9. Elevation of specific cardiac enzymes confirmed the diagnosis of myocardial infarction. True posterior myocardial infarction is difficult to recognise because the leads of the standard 12-lead electrocardiogram are not a direct representation of the area involved. Only with indirect changes in the precordial leads as such the diagnosis can be suspected. This review will highlight the electrocardiographic fine-tuned diagnosis of posterior myocardial infarction by using the posterior leads V7 to V9 leading to easier and faster recognition with consequences for treatment and improved prognosis. (Neth Heart J 2007;15:16-21.)
True posterior myocardial infarction (PMI), the ‘dead angle infarction’ of the electrocardiogram (ECG), is often misjudged and this may be the reason for undertreatment. It is suggested to be one of the most commonly missed types of acute myocardial infarction (MI) electrocardiographic patterns.1 The clinical presentation of PMI is not different from other myocardial infarctions, but the absence of ‘traditional’ electrocardiographic infarct signs such as ST-segment elevation can lead to errors or delay in the diagnosis. Correct interpretation and use of the ECG using the additional leads V7 to V9 can establish the electrocardiographic diagnosis of PMI.
Case report
A 70-year-old woman with a history of thyroid disease, polymyalgia rheumatica and hypercholesterolaemia was admitted to the emergency department. She had complaints of acute chest pain with radiation to the left shoulder, dyspnoea and nausea. On physical examination an adipose woman (BMI 36.2 kg/m2) was seen. Blood pressure was 144/70 mmHg and pulse 77 beats/min. On auscultation normal heart sounds and a midsystolic murmur grade II/VI were heard. Complete physical examination revealed no further abnormalities.
The ECG showed sinus rhythm with a tall R wave in lead V2 , minimal ST-segment elevation in lead III and ST-segment depression in leads V2 to V5, I and aVL (figure 1). There were ST-segment elevations in the posterior leads V7 to V9 (figure 2). Chest X-ray revealed no abnormalities. The patient was at that point in time being treated with a thrombolytic agent because of the clinical and electrocardiographic findings. She was also given aspirin, metoprolol and enoxaparin. She was already taking atorvastatin, levothyroxin and prednisone. Thrombolysis was followed by rapid normalisation of the ST segments (figure 3) and cessation of chest pain. Laboratory results confirmed the diagnosis of MI: troponin-T 0.59 μg/l (N<0.04 μg/l), maximum creatine kinase (CK) 1748 U/l (N=0-200 U/l), and maximum aspartate aminotransferase (ASAT) 199 U/l (N=0-40 U/l). On discharge the patient was on aspirin 80 mg, metoprolol 200 mg, atorvastatin 40 mg, prednisone and levothyroxin. Two months later, the patient was readmitted to the hospital with chest pain. The hospital diagnosis was a non-ST-elevation myocardial infarction (NSTEMI), with troponin-T 0.74 μg/l, maximum CK 1012 U/l and maximum ASAT 134 U/l. Exercise testing before discharge showed significant ST-segment depression suggestive of ischaemia. At coronary angiography (figures 4 and 5) three-vessel coronary artery disease was diagnosed, with an ostial stenosis in the right coronary artery (RCA), a 90% stenosis in the large circumflex artery (RCX) and a 70% stenosis in the left anterior descending artery (LAD). Because of obesity, percutaneous coronary intervention of the RCX and LAD was performed instead of coronary bypass grafting. The mid LAD and proximal RCX were stented. The procedure was uneventful.
12-lead ECG on admission.
Posterior leads V7to V9 on admission.
ECG two days after thrombolysis.
Coronary angiography: right anterior oblique view (left picture) and left anterior view (right picture), left coronary artery, showing a 90% stenosis of the circumflex artery (white arrow) and a 70% stenosis in the left anterior descending (black arrow) artery just distal to a large diagonal branch.
Discussion
The term PMI is used for necrosis of the dorsal, infraatrial part of the left ventricle located beneath the atrioventricular sulcus. The majority of patients with the typical electrocardiographic abnormalities for PMI have a stenosis or occlusion of the RCX. Some patients have a stenosis or occlusion of the RCA. In 10% of the population the RCX is the dominant vessel. It is the least commonly infarcted coronary artery. PMI has been reported to represent 15 to 21% of acute MI, often accompanied by inferior and/or lateral MI. A strict and true PMI is thought to be very rare; however, a more recent study showed an incidence of 3.3% using posterior leads. Risk factors, clinical presentation and differential diagnosis are no different than with other myocardial infarctions.
Rapid recognition of acute PMI is important for several reasons. Patients with inferior or lateral MI who also have PMI have a larger-sized infarct with an increased risk of complications, such as left ventricular dysfunction and death. In a group of 33 consecutive patients with ST-segment elevation only in leads V7 to V9, 22% of patients had moderate to severe mitral regurgitation. Patients with inferoposterior MI may benefit more from reperfusion therapy than patients with myocardial infarction of a ‘single wall’. Patients with electrocardiographic isolated PMI often do not receive the appropriate reperfusion treatment if the clinical diagnosis of MI is not suspected, probably due to lack of the classical ST-segment elevation.
Electrocardiographic findings
Electrocardiographic findings of PMI are shown in table 1. The electrocardiographic diagnosis is difficult because no specific leads of the standard ECG directly represent this area. There is loss of the electrical forces in a dorsal direction, so that a typical infarction pattern only appears in the electrodes placed dorsally between the spine and left scapula on the ECG. On the standard ECG of true PMI the leads V1 and V2 are a mirror image of the V1 and V2 leads of the anterior MI.
Table 1.
| Standard 12-lead ECG |
| ST-segment depression (horizontal >> downsloping/upsloping)* |
| Prominent R wave* |
| R/S wave ratio >1.0 in lead V2 |
| Prominent, upright T wave* |
| Combination of horizontal ST-segment depression with upright T wave* |
| Co-existing acute inferior and/or lateral MI |
| - Additional lead ECG (posterior leads V7 to V9) |
| ≥ 1 mm ST-segment elevation |
* Limited to leads V1 to V3. Adjusted from Brady et al.
QRS complex
During PMI the QRS complex on the vector cardiogram points ventrally due to losses of the electrical forces normally aimed dorsally, resulting in a prolonged R wave. An increase in the R/S ratio >1.0 occurs in leads V1 and V2 as PMI evolves. The increase of the R wave in PMI is the opposite to the Q wave associated with traditional ST-segment elevated myocardial infarction. In some patients the increase of the R wave in lead V1 results from a conduction failure in the His-Purkinje system which is located in the left fascicle of the septum. This suggestion is supported by the fact that about 30% of patients with a PMI develop second- or third-degree atrioventricular block due to ischaemia. A tall R wave in leads V1 and V2 on the ECG is not only seen in right ventricular hypertrophy, hypertrophic cardiomyopathy, right bundle branch block, Wolff-Parkinson-White syndrome, Duchenne muscle dystrophy, right ventricle infarction and posterolateral left wall infarction but also in healthy children and healthy people with prominent forward QRS vectors (table 2). However, with the use of clinical symptoms, additional investigation for these disorders can distinguish PMI.
Table 2.
Differential diagnosis of tall R waves in right precordial leads.
| Diagnosis | Confirmatory clues |
|---|---|
| True posterior infarct | ST↓, T↑ in V1-V2; Q waves and ST↑ V7 to V9 |
| Right ventricular hypertrophy | RAD, RAE; secondary ST-Ts; V7 to V9 normal |
| Ventricular septal hypertrophy | Associated Q waves; LVH; V7 to V9 normal or deep narrow Q waves |
| Right bundle branch block | Wide QRS; broad S in V1, V6; R peaks late in V1; V7 to V9 normal or broad S waves |
| Wolff-Parkinson-White syndrome | Short PR; delta wave; V7 to V9 normal or delta wave |
| Normal variant | No other abnormalities |
LVH=left ventricular hypertrophy RAD=right-axis deviation, RAE=right atrial enlargement. Adjusted from Casas et al.
ST segment
The ST segment points in the direction of the infarcted area and ST-segment depression occurs in the precordial leads in the acute phase. The latter is also seen in precordial ischaemia or as reciprocal changes in MI. The T wave points away from the infarcted area. As a result a forward movement of the T wave can frequently be seen in patients with PMI. The combination of right precordial horizontal ST-segment depression with tall, upright T waves indicates an early electrocardiographic sign of acute ischaemia of the posterior wall during a progressive PMI. The movement of the T wave on the ECG can be ascertained by the T2 to T6 index which is the difference between the voltages of the T waves in leads V2 and V6. If the value of the T2 to T6 index equals or extends the value of 0.38 mV, then the probability of PMI is more than likely.
Use of dorsal leads V7 to V9
Mortality reduction is highest when reperfusion of the infarcted vessel is achieved within six hours of pain onset, with the best results during the first ‘golden’ hour.In search of faster and more reliable methods in identifying PMI, the extra posterior leads V7 to V9 significantly increase the detection of posterior injury patterns compared with the standard 12-lead ECG. Lead V7 should be placed at the level of lead V6 at the posterior axillary line, lead V8 on the left side of the back at the tip of the scapula and lead V9 is placed halfway between lead V9 and the left paraspinal muscles. ST-segment elevation of >1 mm in the posterior leads is suggestive of PMI. Using all 15 leads significantly improves the further detection of circumflex coronaryrelated injury pattern over the standard 12-lead ECG. Sensitivity increased from 32 to 57% with a specificity of 98% for the circumflex artery. Five percent of diagnosed non-Q-wave MIs are retrospectively PMIs in which reperfusion therapy would have been justified if posterior leads had been recorded. The sensitivity and positive predictive value for the diagnosis of MI with the additional leads V7 to V9 and right ventricular leads V4R to V6R increases from 57.7 to 66.1% and from 88.4 to 96.8%, respectively, but the specificity for MI decreases from 91 to 84%. The overall accuracy of the additional-lead ECG is only modestly improved.6 A study investigating the use of 15-lead ECG in comparison with 12-lead ECG in every emergency chest pain patient showed no alterations in diagnosis and management. It is advised to only use the 15-lead ECG in a subgroup of patients more likely to have PMI or right ventricular infarction. There is an increase in reported incidence of PMI using the 15-lead ECG compared with the 12-lead ECG, suggesting more undiagnosed PMI using the 12-lead ECG.20 Kulkarni et al. prospectively showed the electrocardiographic changes during RCA and RCX balloon inflation, mimicking an occlusion in the related artery.21 They found that ST-segment elevation restricted to V7 to V9 was only seen during RCX inflation. Inferior elevation alone was not seen with RCX inflation. Patients with ST-segment elevation in the inferior and posterior leads are more likely to have RCX occlusion rather than RCA (85%). Schmitt et al. found the sensitivity of the ECG diagnosis of AMI was increased by 11% (from 50 to 61%) for RCX lesions by extended precordial leads, after comparing ECG diagnosis with angiographic findings. They found a trend toward an extended infarct size in those patients with concomitant ST-segment elevation in the additional ECG leads.
There are no studies showing improved outcome after thrombolysis in PMI. Thrombolysis has potential hazardous complications so there should be awareness for pseudo infarct patterns. As with any MI it is important to start reperfusion treatment as soon as possible when there is a strong suspicion for PMI based on clinical signs, symptoms and electrocardiographic findings.
In the early stage of acute MI the ECG may be normal or near normal. Less than 50% of patients with acute MI have clear changes suggestive of the diagnosis on their first ECG. About 10% of laboratory-proven MIs are NSTEMI. Thrombolysis is justified in patients with >1 mm ST elevation in two contiguous leads in the limb leads or >2 mm ST elevation in two contiguous chest leads or in patients with new left bundle branch block. Fibrinolytic therapy may be appropriate when there is marked ST-segment depression confined to leads V1 to V4 accompanied by tall R waves in the right precordial leads and upright T waves indicative of true posterior injury. Confirmatory data from posterior leads may be especially helpful. It is suggested that because of the greater distance between the infarcted area and the leads in PMI, an elevation of 0.5 mm is sufficient to justify the diagnosis of PMI followed by decisions in reperfusion treatment. Adjusted criteria provided an improved sensitivity from 49% in the 12-lead to 94% in 15-lead ECG. An increase of the use of thrombolysis was seen with the increasing number of electrocardiographic leads demonstrating ST-segment elevation. Posterior chest leads should be routinely recorded in patients with suspected MI and nondiagnostic routine ECG to establish the appropriate reperfusion treatment (including thrombolysis) of some of the patients now classified as having unstable or non-Q-wave MI. Some authors claim that in specific cases ST-segment depression in the precordial leads is sufficient for thrombolysis.28 By using the posterior leads V7 to V9 this uncertainty will not be necessary in most cases. Primary angioplasty has the advantage of establishing the diagnosis and therapy immediately. It should be emphasised that patients presenting with MI with co-existing PMI are at greater risk of complications, and acute therapy including thrombolysis and angioplasty should not be delayed.
Conclusion
PMI is responsible for subtle changes on the ECG. For the diagnosis of PMI it is important to recognise the clinical signs at presentation, combined with subtle manifestations on the ECG in order to start reperfusion therapy early. Cardiologists should be able to interpret electrocardiographic signs such as ST-segment depression with upright T waves and prominent tall R waves in leads V1 to V3 in order to make sure that a posterior lead ECG is recorded in these cases. Using posterior leads in patients presenting with symptoms suspicious for MI will reveal more patients with PMI who will benefit from early reperfusion treatment. Retrospective analysis of available data concerning non-ST-segment ASC might give new insights in the indication of PMI.
